Co-authored by: Benedikt Marijnen, LLM – European Lawyer In the past year I’ve spoken to hospital 3D lab managers across Europe who were operating under the assumption that Article 5.5 of EU MDR covered their work. It’s an understandable assumption. Article 5.5 is the provision that gives health institutions some breathing room from the full …
Category: KnowledgeBase
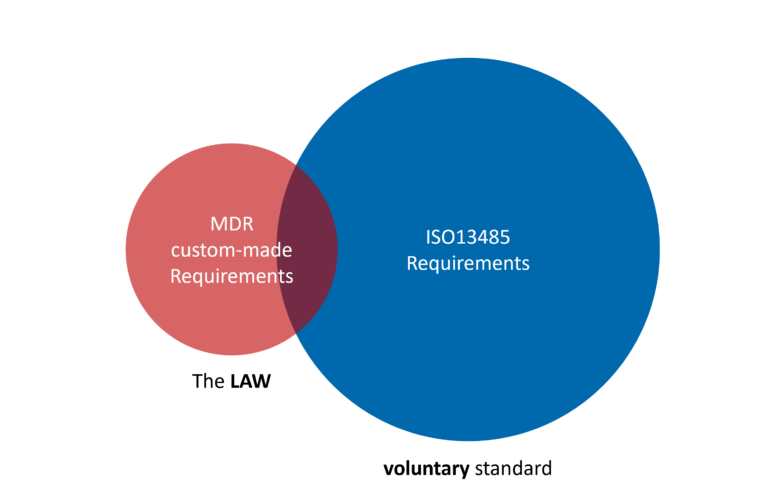
Is ISO 13485 actually necessary for hospital 3D labs?
Many hospital 3D labs I speak with run into the same situation. They start looking into the Medical Device Regulation (MDR) and quickly hear the same advice: “You need an ISO 13485 Quality Management System.” ISO 13485 is indeed a well-established standard for medical device manufacturers. But for hospital-based 3D labs producing custom-made devices, the …
Safety and performance
All medical devices, including personalized/custom-made (patient-matched) devices, devices made under the healthcare institution exemption (point-of-care manufacturing) and 3D printed medical devices have to adhere to the applicable parts of Annex I of the MDR. This IMDRF/GRRP WG/N47 FINAL:2024 (Edition 2) guidance document might help to make sense of the Essential Principles of Safetyand Performance:
3D printing at Point-of-Care
3D technology has found it’s way to the hospital and is significantly improving patient care. In the article below I highlight the opportunities and identify the risks of 3D printing in a hospital setting. It talks about regulatory and quality management aspects and links to helpful guidance documents.
Procedures and form templates
Struggling with a certain Procedure (SOP) or Form templates in your ISO 13485-based QMS? Wouldn’t it be nice to get that documented procedure from a certified QMS that has stood the test of several Notified Body audits (and multiple continuous improvement iterations)? Via the form below you can request to receive one or more stand-alone …
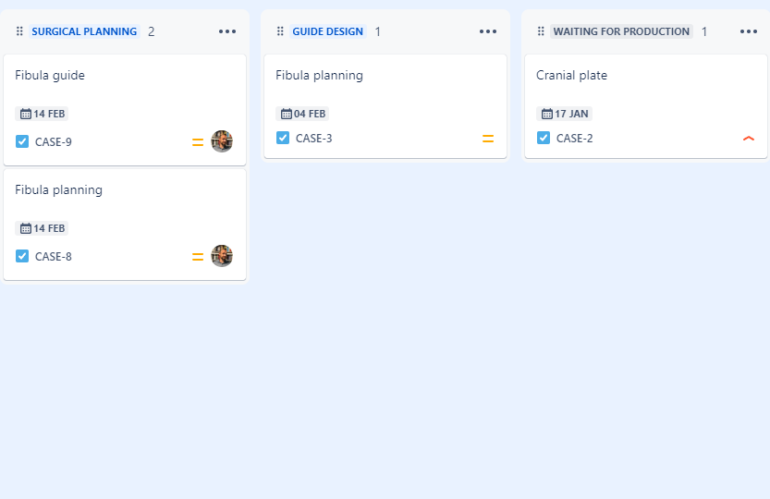
Case management in personalized medical devices
Case Management in Patient-Specific Surgical Planning, Surgical Guides, and Custom-Made Implants Case management plays a vital role in the production and implementation of 3D-printed patient-specific surgical planning, surgical guides, and custom-made implants. Not only does it ensure streamlined operations, but it also helps achieve traceability—a fundamental requirement of a Quality Management System (QMS) under ISO …
Risks in medical 3D printing
Risks are everywhere! As part of your QMS, analyse your risks. It’s convenient to analyse the risks per step in the workflow below. Do this with a multidisciplinary team, so you can approach this from various angles. This workflow reflects the elements of the Ishikawa diagram, or 5M method: Sometimes you even see another M: …
The best software for QMS…
…is not QMS software (and is affordable!) In this whitepaper I share the lessons learned while looking to implement an ISO13485 Quality Management System for a medical device company that 3D prints custom-made medical devices and that have proven to be applicable to other medical devices.
Practical guide to ISO 13485
Where to start with your QMS for ISO 13485? Struggling to start with your QMS? In this practical guide I share my proven approach in setting up and implementing an (ISO13485-based) Quality Management System for a (3D printing) medical device company or Point-of-Care 3D Lab.
Build a digital QMS for ISO13485…
…without QMS software Most people who want to start building a Quality Management System (QMS) look at dedicated QMS software first. So did I years ago. But after a thorough “Quest for Quality” I chose a more efficient ánd economical approach. I wrote a whitepaper to share the lessons learned while looking to implement an …