Co-authored by: Benedikt Marijnen, LLM – European Lawyer In the past year I’ve spoken to hospital 3D lab managers across Europe who were operating under the assumption that Article 5.5 of EU MDR covered their work. It’s an understandable assumption. Article 5.5 is the provision that gives health institutions some breathing room from the full …
Tag: MDR
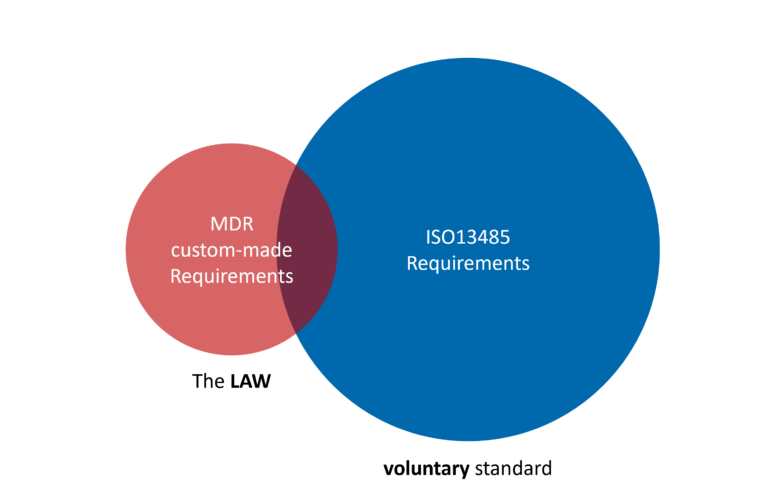
Is ISO 13485 actually necessary for hospital 3D labs?
Many hospital 3D labs I speak with run into the same situation. They start looking into the Medical Device Regulation (MDR) and quickly hear the same advice: “You need an ISO 13485 Quality Management System.” ISO 13485 is indeed a well-established standard for medical device manufacturers. But for hospital-based 3D labs producing custom-made devices, the …