Many hospital 3D labs I speak with run into the same situation.
They start looking into the Medical Device Regulation (MDR) and quickly hear the same advice:
“You need an ISO 13485 Quality Management System.”
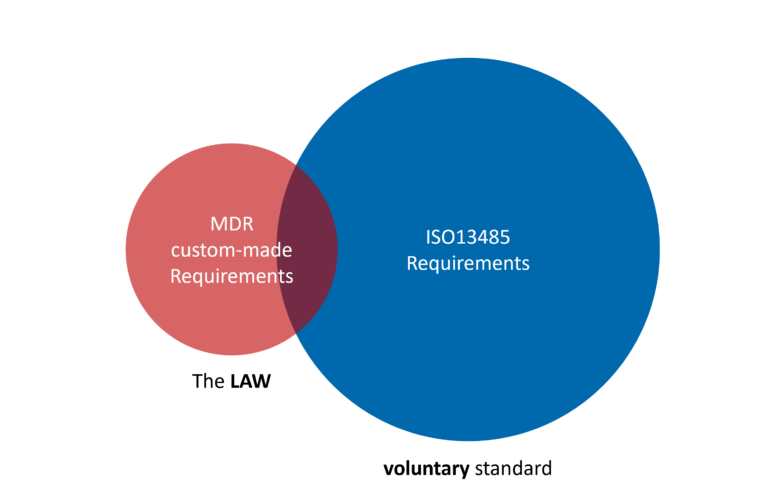
ISO 13485 is indeed a well-established standard for medical device manufacturers. But for hospital-based 3D labs producing custom-made devices, the picture is a bit more nuanced.
ISO 13485 was written for traditional manufacturers
ISO 13485 was primarily written for companies that:
- design standardized medical devices
- manufacture them in series
- place them on the market
- distribute them internationally
Think of companies producing implants or surgical instruments at scale.
Hospital 3D labs operate in a very different environment.
Most hospital labs produce patient-specific or custom-made devices, often:
- based on CT or MRI data
- produced for a single patient
- manufactured in very small quantities
- used within the same hospital
That difference has important implications for compliance.
ISO 13485 can quickly become heavy for small labs
A full ISO 13485 system includes many elements that are primarily relevant for commercial manufacturers with complex supply chains and distribution networks.
Examples include:
- extensive supplier qualification
- thorough human resource procedures
- complex purchasing controls
- procedure for internal audits
For a small hospital lab producing a limited number of patient-specific devices, implementing all of this can quickly become disproportionate.
In practice, many labs spend months building documentation that plays only a limited role in their daily workflow.
ISO 13485 does not automatically ensure MDR compliance
Another common misunderstanding is that ISO 13485 automatically ensures MDR compliance.
In reality, the MDR contains several requirements that are not explicitly covered by the ISO standard, especially for custom-made devices.
Examples include:
- the custom-made device statement required under MDR Annex XIII
- the formal role of the prescribing healthcare professional
- clinical evaluation for the device family
This means that even a well-implemented ISO 13485 system may still require additional work to fully comply with the MDR.
MDR and ISO 13485: overlapping but not identical


The overlap between ISO 13485 and the MDR includes many familiar quality management elements such as:
- risk management
- traceability
- corrective actions
But both frameworks also contain requirements that do not fully overlap.
For hospital 3D labs producing custom-made devices, this difference can be important when deciding how to structure compliance efforts.
3 MDR pitfalls I often see in hospital 3D labs
When speaking with hospital 3D labs across Europe, I notice that many teams run into similar challenges when trying to implement the MDR.
Not because they don’t take compliance seriously; quite the opposite.
But because the regulation was not originally written with small hospital innovation labs in mind.
1. Assuming supplier certificates solve compliance
A common assumption is that using:
- certified software
- biocompatible materials
- certified 3D printers
automatically ensures MDR compliance.
But under the MDR, responsibility always remains with the manufacturer of the final device.
Supplier documentation can support your case, but it does not replace:
- risk management
- validation of the manufacturing process
- verification/validation of the final device
In other words:
Compliance lives in the process, not in the components.
2. Confusing dimensional accuracy with device quality
In 3D printing environments, quality is often equated with dimensional accuracy. If the printed device matches the digital design within tolerance, it is considered “good”.
However, under the MDR, quality goes far beyond geometry.
A device can be perfectly dimensionally accurate and still be unsafe or non-compliant. For example if:
- uncured resin residues remain on the surface
- the device has not been properly cleaned or post-processed
- it has weak parts that are mechanically unsuitable for the intended use
In other words:
A perfect shape does not automatically mean a safe medical device.
3. Starting with ISO 13485 before understanding MDR obligations
ISO 13485 can be a powerful framework.
But many hospital labs start building a full ISO system before clearly mapping their MDR obligations.
This often leads to months of work on procedures that are not directly relevant for their situation.
A more effective approach is often:
- understand the MDR obligations for custom-made devices
- implement the required documentation and processes
- then decide whether a full ISO 13485 system is necessary
For many hospital labs, that sequence turns out to be much more manageable.
When ISO 13485 does make sense
None of this means ISO 13485 is irrelevant.
On the contrary: it becomes increasingly valuable when a lab grows or its role changes.
For example:
- when a hospital lab starts supplying devices to other hospitals
- when production scales up significantly
- when devices move from custom-made to standardized products
- when 3D labs want to demonstrate a mature quality system to partners or regulators
At that stage, ISO 13485 provides a strong and internationally recognized framework.
The real question
Hospital 3D labs are doing incredible work bringing patient-specific innovation directly into clinical practice.
The challenge is making sure the regulatory framework supports that innovation, rather than slowing it down unnecessarily.
For many labs, the real question is not:
“How do we implement ISO 13485?”
But rather:
“What is the most practical way for a hospital lab to comply with the MDR?”
Over the past year I’ve been exploring ways to make MDR compliance more practical for hospital 3D labs, with a reduced QMS, based on ISO13485.
One interesting development is the emergence of a structured MDR compliance platform, developed by legal experts specialized in medical device regulation, that translate the regulation directly into practical documentation and workflows.
It’s encouraging to see solutions emerging that are better aligned with how hospital innovation labs actually operate.
Because ultimately, when innovation meets compliance, it becomes patient care!