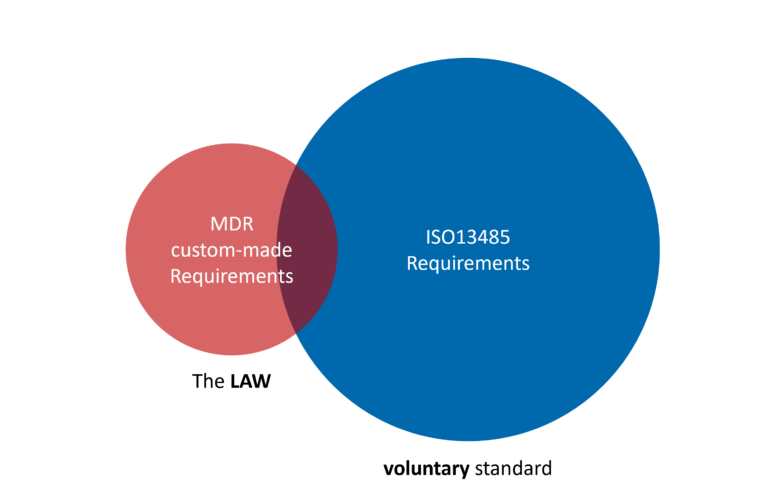
Many hospital 3D labs I speak with run into the same situation. They start looking into the Medical Device Regulation (MDR) and quickly hear the same advice: “You need an ISO 13485 Quality Management System.” ISO 13485 is indeed a well-established standard for medical device manufacturers. But for hospital-based 3D labs producing custom-made devices, the …
Tag: medical devices
Safety and performance
All medical devices, including personalized/custom-made (patient-matched) devices, devices made under the healthcare institution exemption (point-of-care manufacturing) and 3D printed medical devices have to adhere to the applicable parts of Annex I of the MDR. This IMDRF/GRRP WG/N47 FINAL:2024 (Edition 2) guidance document might help to make sense of the Essential Principles of Safetyand Performance:
Guidance PMD
Personalized Medical Devices – Regulatory Pathways Last week, the IMDRF released the second edition (2023) of document IMDRF/PMD WG/N58. The previous edition dated from 2020. In this latest version, they elaborate a bit more on the concept of the Medical Device Production System (MDPS).